|
10/2/2023 0 Comments No molecular orbital diagramNitrogen atom has 2p atomic orbitals lower by 2.52 eV, and 2s atomic orbitals lower by 6.13 eV than with carbon atom. Thus, simply add one or two electrons into the 2b3u and 2b2u to get NO2 and NO 2, respectively. The wave function describes the wavelike properties of an electron. You've seen the molecular orbital (MO) diagram of CO2: CO2 and NO+ 2 are isoelectronic and thus have the same electron configuration. The mathematical process of combining atomic orbitals to generate molecular orbitals is called the linear combination of atomic orbitals (LCAO). In these diatomic molecules, several types of molecular orbitals occur. Number of electrons present in the bonding orbitals is represented by Nb and the. Such molecules are called homonuclear diatomic molecules. 1) Stability of molecules in terms of bonding and antibonding electrons. We will consider the molecular orbitals in molecules composed of two identical atoms (H 2 or Cl 2, for example). Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin. The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Needs multiple structures to describe resonance An honors general chemistry computational lab as implemented using three dimensional modeling software journal of chemical education ruddick parrill and petersen 2012 89 11 pp 13581363 abstract. These are uvvisible infra red ir and nuclear magnetic resonance nmr spectroscopies. Predicts the arrangement of electrons in molecules Draw the molecular orbital diagrams for no and no. Predicts molecular shape based on the number of regions of electron density And, we need to learn both in an extensive format to grasp the reality of bonding nature inside any molecule. Hybridization and Molecular Orbital theory are two concepts that quite overlap each other. 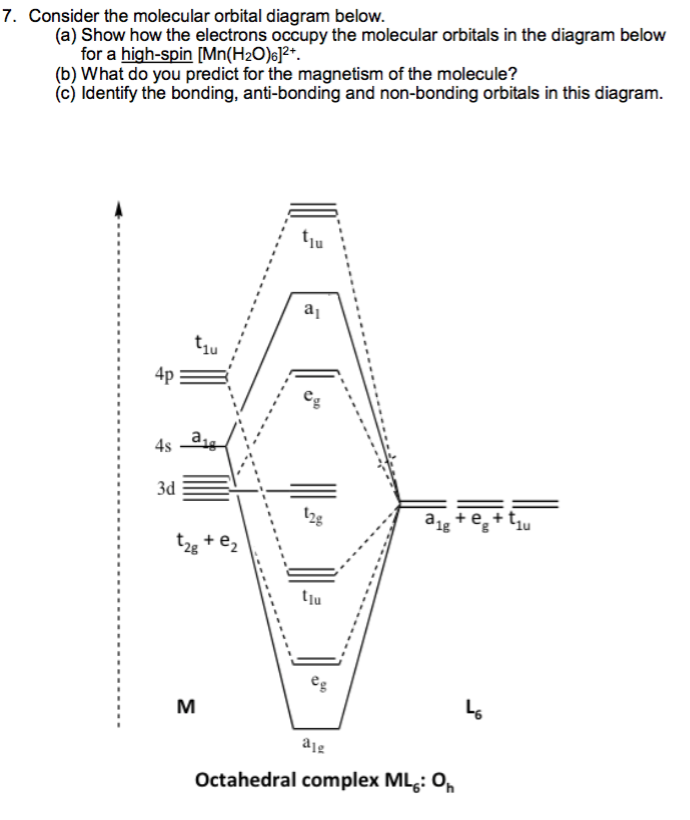 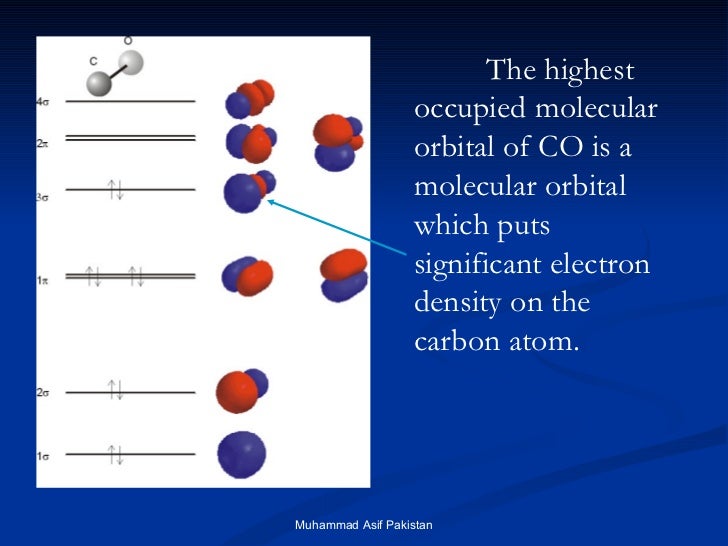
\) : Comparison of Bonding Theories Valence Bond TheoryĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled NO Hybridization and Molecular Orbital (MO) Diagram.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
